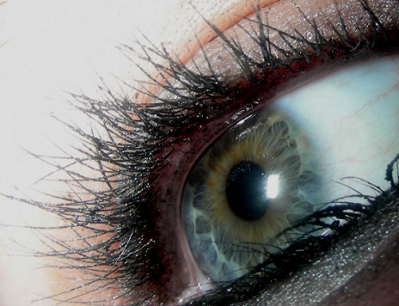
Eyelash enhancer avoids regulatory trap
landing two companies in a regulatory snare but La Canada Ventures
has been careful to avoid trouble.
Rivals withdraw products Both Jan Marini Skin Research and Athena were forced to withdraw their eyelash enhancing products because they contained bimatoprost, an ingredient licensed for use in a glaucoma drug.
In November the FDA seized around $2m worth of Jan Marini's Age Intervention Eyelash Enhancer calling it an unapproved and misbranded drug because it contained this ingredient and claimed to actually increase eyelash growth.
Days later Athena voluntarily withdrew its Revitalash product citing 'possible public concern over bimatoprost'.
Meanwhile, one of their closest competitors, La Canada Ventures, has avoided any regulatory difficulties with its eyelash conditioner MD Lash Factor, which was launched six months ago.
Following guidelines The president of La Canada Ventures, Susan Lin, said the company has escaped conflict with the FDA because it has followed the rules and clearly marketed its product as a cosmetic.
Firstly, the active ingredient in MD Lash Factor is not used in a drug but is a newly patented substance called dechloro ethylcloprostenolamide.
To make doubly sure that the ingredient would not land the company in trouble, it conducted extensive in-vitro and in-vivo tests and submitted the positive results to the FDA.
Secondly, La Canada Ventures does not claim that MD Lash Factor increases eyelash growth but that it improves the appearance of natural lash length classifying it as a cosmetic rather than a drug.
Susan Lin said: "I am surprised about how others didn't follow the FDA rules."
Product claims While La Canada Ventures does not market MD Lash Factor as a product that increases eyelash growth, the company has tested its effectiveness and claims that it is a breakthrough technology.
The company claimed that a randomized double blind study with 34 volunteers on the eyelash conditioner indicates a 53 percent increase in length after four weeks of daily use.
In its marketing La Canada Ventures is faced with the challenge of persuading consumers that its product is a major step forward without saying that it actually causes the eyelashes to grow.
Before and after photographs are used in its advertising to indicate how effective it is and avoid any semantic traps.
The product is currently being sold online and through physicians offices in an effort to establish it in the medical market and give it credence.
It is currently available in the US and several other countries including Australia, Canada and France with plans to take the product into other markets.
Lin said the market was being sold as a cosmetic in these foreign markets although how the regulatory differences affect the positioning of the product is a question that needs to be resolved.
Susan Lin will be presenting the MD Lash Factor at the HBA Spring Conference in New York next month in a talk on promising new technologies entitled "Great Expectations: Eyelash Enhancement Properties of Topical Dechloro Ethylcloprostenolamide" .

![Indus Valley is working to corner 30% of India's online premium boxed hair colour market. [Indus Valley]](/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/article/2024/07/26/indus-valley-aims-to-secure-30-of-india-s-online-premium-hair-colour-market-with-organic-offerings/17594932-5-eng-GB/Indus-Valley-aims-to-secure-30-of-India-s-online-premium-hair-colour-market-with-organic-offerings.jpg)
![[Getty Images]](/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/china/china-focus-latest-developments-in-china-s-booming-beauty-market25/17606695-1-eng-GB/China-focus-Latest-developments-in-China-s-booming-beauty-market.jpg)
![Kosé has launched makeup brand Visée in Singapore as part of plans to reinforce its position in SEA. [Visée]](/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/headlines/business-financial/visee-singapore-kose-aims-to-enhance-brand-visibility-in-sea-with-new-launch/17587264-1-eng-GB/Visee-Singapore-Kose-aims-to-enhance-brand-visibility-in-SEA-with-new-launch.jpg)
![ble C&C is set on reinforcing its competitiveness in China’s beauty market. [Missha]](/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/headlines/business-financial/able-c-c-aims-to-strengthen-competitiveness-in-china-through-online-expansion-kol-collabs/17591626-1-eng-GB/Able-C-C-aims-to-strengthen-competitiveness-in-China-through-online-expansion-KOL-collabs.jpg)

![LG H&H genetic study says 23 genetic regions affect natural skin tone. [Getty Images]](/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/article/2024/07/23/lg-h-h-discovery-of-genetic-skin-tone-factors-in-east-asians-potentially-key-to-skin-radiance-developments/17587210-1-eng-GB/LG-H-H-discovery-of-genetic-skin-tone-factors-in-East-Asians-potentially-key-to-skin-radiance-developments.jpg)

![DR.CI:LABO expects brand-supplier partnerships gain more public prominence as consumers interest in skin care grows online. [Dr.Ci:Labo]](/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/article/2024/07/22/brand-supplier-partnerships-will-come-to-the-fore-amid-the-online-skin-care-landscape-dr.ci-labo/17576755-1-eng-GB/Brand-supplier-partnerships-will-come-to-the-fore-amid-the-online-skin-care-landscape-DR.CI-LABO.png)